 In ionic and covalent materials, solids result from strong ionic bonds or the intermolecular forces between molecules. In chemistry class, you probably focused on molecular and ionic bonds, in which electrons are shared to varying degrees between individual atoms. How is it possible for metals to flex in so many different ways? The answer is metallic bonding. They can be formed into microcircuits for cell phones, or used in the development of smart coatings for glass that not only make windows reflective but also protective by blocking infrared radiation. While aluminum foil in your kitchen and copper electrical wires in electric cables are obvious everyday uses of these properties, wires can be shrunk down in complex ways. They are also ductile, which means they can be easily drawn into wires. Metals are malleable, meaning that they can be formed into other shapes, such as thin sheets or foils, without breaking or cracking. But do you know why? What makes metals so marvelous? The answer, of course, lies at the atomic scale. They are shiny, malleable, fusible, ductile, good conductors of electricity and heat, and inclined to give up electrons to form cations. You already know some technical basics about metals. From gold nanoparticles that can detect and treat cancer to vanadium-stabilized batteries to aluminum-based materials that can harvest water from desert air, metals are humanity’s past, present, and future. Not to mention the copper, silicon, and rare-earth metals that make all of our electrical power, computers, and smart devices possible.Īnd the possibilities keep growing. Aluminum makes aviation possible, and steel (mostly iron) forms the frames of cars, buses, and trains. 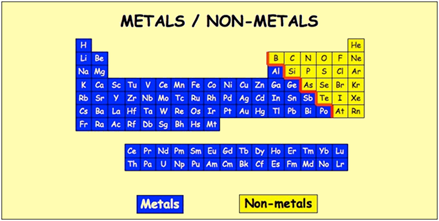
Want to get from city to city or around the world without metals-forget it. The cobalt in vitamin B12 helps us make red blood cells, the iron in hemoglobin carries oxygen to tissues, and sodium and potassium ions allow the heart and nerves to communicate. These ubiquitous elements, which make up more than 75% of the periodic table, are everywhere doing amazing things. Such a classification should reflect our understanding of the chemical basis of toxicity and allow toxic effects to be predicted.The 2019 National Chemistry Week (October 20-26) theme is “Marvelous Metals,” and no wonder. In order to avoid the use of the term "heavy metal", a new classification based on the periodic table is needed. This usage implies that the pure metal and all its compounds have the same physicochemical, biological, and toxicological properties, which is untrue. Even the term "metal" is commonly misused in both toxicological literature and in legislation to mean the pure metal and all the chemical species in which it may exist. Thus, the term "heavy metals" is both meaningless and misleading. 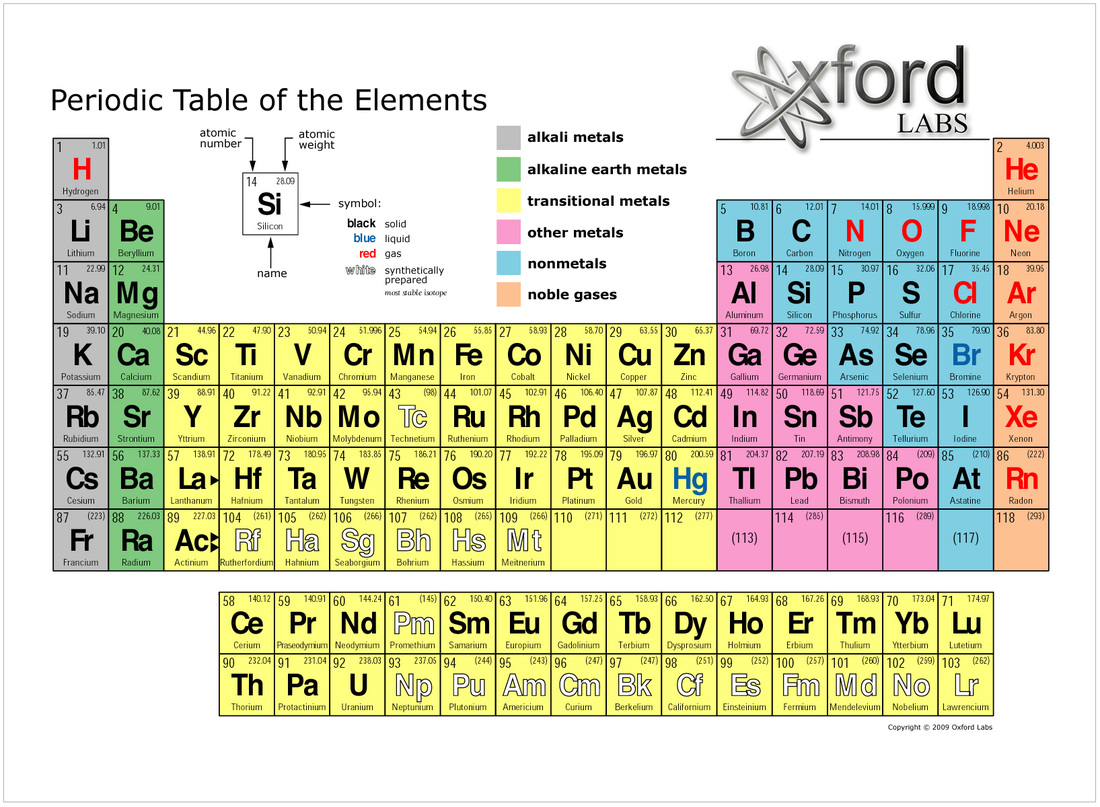
This has no basis in chemical or toxicological data. There is a tendency, unsupported by the facts, to assume that all so-called "heavy metals" and their compounds have highly toxic or ecotoxic properties. However, there is no authoritative definition to be found in the relevant literature. Such lists differ from one set of regulations to another and the term is sometimes used without even specifying which "heavy metals" are covered. At the same time, legal regulations often specify a list of "heavy metals" to which they apply. It is often used as a group name for metals and semimetals (metalloids) that have been associated with contamination and potential toxicity or ecotoxicity. Over the past two decades, the term "heavy metals" has been widely used. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
